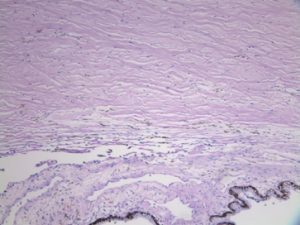
Glaucoma is a leading cause of irreversible blindness in people and animals worldwide that results from loss of neurons in the optic nerve and retina. In many cases, disruption of normal pathways for outflow of aqueous humor (the fluid that circulates and drains within the anterior chamber of the eye) results in an intraocular pressure (IOP) that is too high and that has a negative impact on the health and function of the optic nerve and retina. Our research program focuses on the processes involved in optic nerve axon loss in glaucoma and factors that determine susceptibility to loss of vision in affected individuals. An important goal of our current research is to accelerate translation of promising new strategies to preserve vision in patients diagnosed with glaucoma.
The McLellan Lab is well equipped to conduct complex independent and collaborative basic research projects spanning gene and protein expression studies, cell biology, pathology, in vivo imaging and electrophysiology, with a focus on type 1 translational research. We have expertise in assessment of visual function, comparative ocular pathology and advanced imaging of the eye in a range of species.
Below are the current initiatives for our lab.
Research Areas
Targeting fibrosis and gliosis in the optic nerve head
Currently, treatment for glaucoma patients hinges on medical and surgical strategies to lower intraocular pressure (IOP). However, progressive loss of vision occurs in a significant proportion of glaucoma patients despite seemingly adequate regulation of IOP. Prevention of adverse scar formation, neuroinflammation and nerve fiber loss in the optic nerve of patients with glaucoma remains an unmet clinical goal. Transforming growth factor-β (TGF-β) has been widely implicated as playing a significant role in scarring and extracellular matrix tissue remodeling in optic nerve head tissues (from human donors, veterinary patients and experimental animal models) and in cultured optic nerve head cells in vitro. The TGF-β signaling pathway is therefore considered as a promising “druggable” target for therapy in glaucoma patients.
One objective of current research in our lab is to modulate tissue TGB-β expression and signaling, and limit scarring and damage to the optic nerve by repurposing an existing “multifunctional” drug to complement existing glaucoma treatments. Our studies examine the effects of this drug on the complex population of optic nerve head cells and gene expression in situ and vitro , while determining its therapeutic benefits in vivo, within a clinically relevant treatment window in a clinically relevant model. Our work is set to increase the overall understanding of the role of TGF-β in glaucomatous optic neuropathy and extracellular matrix disorders in human and animal patients.
Aqueous Humor Outflow Pathways in Pediatric Glaucoma
Glaucoma is widely recognized as a common, blinding disease affecting older adults, but glaucoma is also an important cause of vision loss in children world-wide. Primary congenital glaucoma (PCG) is relatively rare with an estimated incidence of around 1:20,000 live births in developed Western countries. However, the disease has genetic basis in most cases and incidence can be as high as 1:1,250 live births in some populations. Rigorous, ongoing medical therapy and multiple surgical procedures are necessary to provide any hope of preserving lifetime vision. Unfortunately, these demanding treatments can take a heavy toll on overall quality of life for affected children and their families.
Intraocular pressure (IOP) is a consistent risk factor for vision loss in glaucoma. Maintenance of a normal IOP requires balance between aqueous humor production and outflow from the anterior chamber. Abnormal or arrested development of vascular channels that serve as a pathway for outflow of aqueous humor results in an unhealthy build-up of IOP causing congenital and pediatric glaucoma.
In the absence of rational and effective treatments that directly and specifically address the unique underlying structural abnormalities in children with glaucoma, the goal of eliminating discomfort and preserving life-time vision can sadly prove elusive for some pediatric glaucoma patients. Studies currently underway in our lab are aimed at improving understanding of how the normal vascular channels responsible for outflow of aqueous humor from the eye develop in a broad range of animal species using cutting edge imaging tools to visualize these pathways in live patients and by adopting exciting new microscopy techniques to visualize these pathways in pathology specimens. Together with our collaborators, including Dr Terri Young, Dr Yasmin Bradfield and the UW team of pediatric ophthalmologists and glaucoma specialists, our long-range goal is to develop and test new, complementary treatment strategies that will ultimately help children with PCG maintain life-long vision. As a step in this process our lab is working to help identify key biologic molecules and pathways responsible for early-life development of the aqueous outflow vessels.
Animal Models: Role in Fighting Blindness in Human and Veterinary Patients
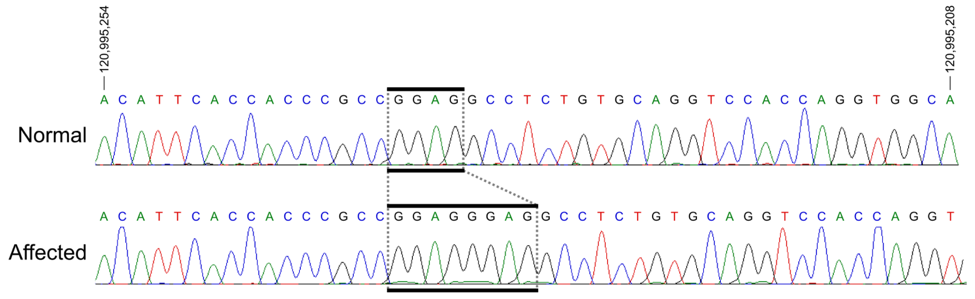
A significant strength of our program is the unique, feline genetic model of spontaneous glaucoma that we have extensively characterized. Working with our collaborators, we identified a genetic mutation responsible for development of congenital glaucoma in cats. The gene involved, LTBP2, also causes glaucoma in humans. Like humans, the cat optic nerve head has a well-developed, collagenous lamina cribrosa (LC); onset of glaucoma occurs early in life, and slow disease progression provides an extended “window” in which to study treatment effects.
Our ongoing studies in this model aim to provide proof of concept that novel treatment strategies can modify disease progression in glaucoma to preserve vision. By repurposing existing FDA approved drugs with established safety profiles, and testing these treatments in a spontaneous animal model under clinically relevant conditions, our approach will inform clinical trial design and accelerate translation to human and animal glaucoma patients.
Our studies will suggest novel approaches to prolong function and survival of cells in the retina and optic nerve in pediatric glaucoma. Our contributions will also have broader impact and translational significance by suggesting target mechanisms to limit pathological processes common to glaucoma and other debilitating central nervous system injuries and neurodegenerative diseases.